Testing for Novel Corona virus (SARS-CoV-2) is done by detection Viral RNA detection by PCR Techniques
Existing Multiplex PCR kits with respiratory virus panels, such as those manufactured by Biofire or Genmark, DO NOT detect CoV-2, the virus that causes COVID-19.
Lower respiratory specimens likely have a higher diagnostic value than upper respiratory tract specimens for detecting COVID-19 infection. WHO recommends that, if possible, lower respiratory specimens such as sputum, endotracheal aspirate, or bronchoalveolar lavage be collected for COVID-19 testing. If patients do not have signs or symptoms of lower respiratory tract disease or specimen collection for lower respiratory tract disease is clinically indicated but the collection is not possible, upper respiratory tract specimens, such as a nasopharyngeal aspirate or combined nasopharyngeal and oropharyngeal swabs should be collected.
Serological testing is not available currently, when it becomes available: serum, acute and convalescent (possibly 2-4 weeks after acute phase) can be considered.
In India, National Institute of Virology, Pune, of the Indian Council of Medical Research (ICMR) is the apex laboratory for reconfirmation of any positive samples as well as for the quality assurance of the testing of samples for COVID-19. In addition, 13 Viral Research and Diagnostic Laboratories (VRDLs) and National Centre for Disease Control (NCDC), New Delhi are performing tests for the COVID-19.
In Delhi, a 24×7 National Control Room (011-23978046) has been made operational.
CLICK Here for Details issued by WHO about laboratory guidance for COVID-19.
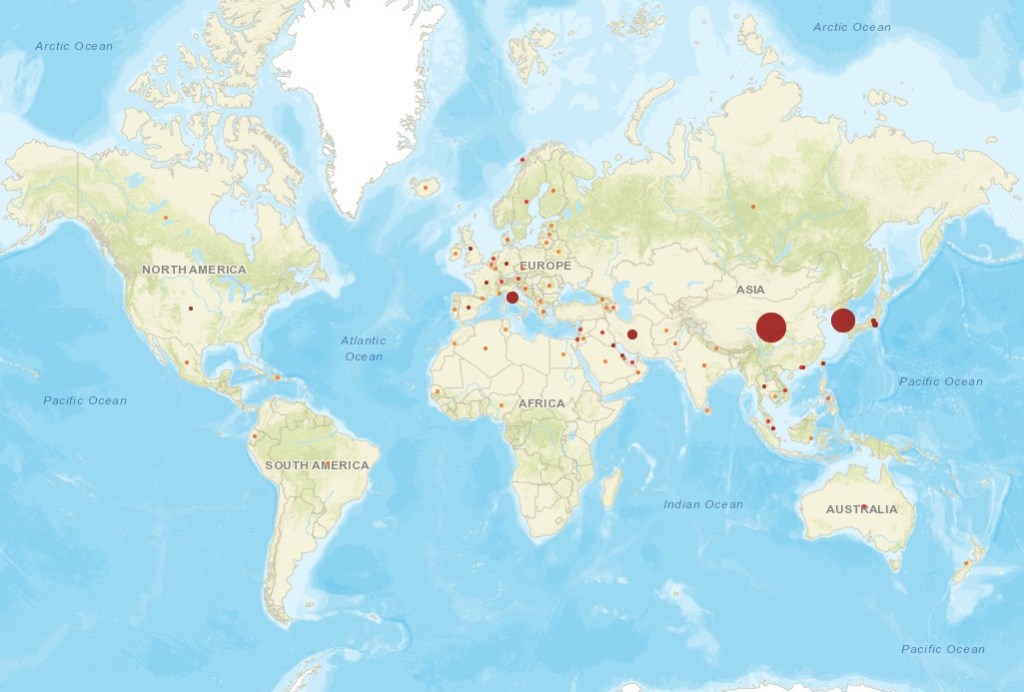
CLICK BELOW TO SEE LIVE STATUS OF COVID-19 CASES REPORTED TILL DATE